Controls for antibody cross reactivity1/17/2024  
Consequently, they argue that the induction of a broad immune response offers the prospect of preemptive vaccine updates (12, 14). Here, the authors argue that this “backboost” has a positive contribution to vaccine efficacy by helping maintain immunity to a broad range of influenza viruses. As an example, Fonville and others observed that influenza virus infections not only induce new antibodies targeting the current infection or vaccination strain but also boost antibody titers against a broad range of preceding heterologous influenza virus infections and vaccinations (12, 13). Memory recall of B cells has been related to both positive and negative outcomes of heterologous virus infections. Immunity to antigenically related pathogens affects the development of a new immune response and is a key factor in the clinical outcome of infection (11). Thus, there is an urgent need for detailed insight into the SARS-CoV-2 immune response in the context of a CCC-experienced immune system. A lack of knowledge regarding the specific effector mechanisms associated with protection against SARS-CoV-2 in COVID-19 hampers the development of targeted immune modulators to prevent or overcome severe disease (10). However, preexisting immunity may also promote pathology (9). Preexisting memory B cells that were once primed by antigenically related seasonal common cold coronaviruses (CCCs) may provide fast protection against SARS-CoV-2 infection by a rapid production of cross-reactive antibodies from memory recall, e.g., cross-neutralizing antibodies (5, 8). The kinetics of preexisting and newly induced antibodies upon SARS-CoV-2 infection are expected to be important. Some patients with COVID-19 develop acute respiratory distress syndrome (ARDS) that requires treatment in an intensive care unit (ICU) and carries in a high mortality rate (2–4).Īlthough correlates of protection against severe COVID-19 are not fully defined in humans, SARS-CoV-2–neutralizing antibodies are considered a hallmark of immune protection (5–7). The majority of patients with COVID-19 experience mild symptoms including fever, cough, and myalgia, none of which can be considered specific to SARS-CoV-2 infection (2). 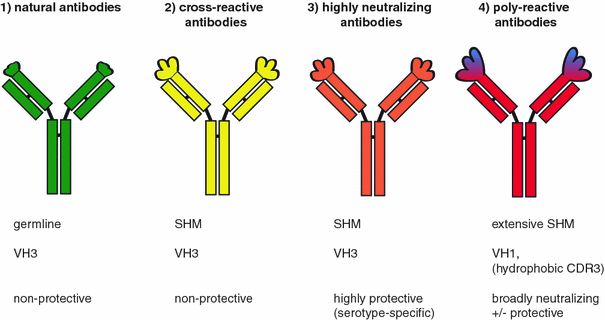
The introduction of the zoonotic severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has led to a pandemic of coronavirus disease 2019 (COVID-19) (1). These findings indicate a boost of poorly protective CoV-specific antibodies in patients with COVID-19 that correlated with disease severity, revealing “original antigenic sin.” Graphical Abstract These boosted clones showed limited cross-reactivity and did not neutralize SARS-CoV-2. Additionally, IgG clones directed against a seasonal coronavirus were boosted in patients with severe COVID-19. Patients with COVID-19 mounted a mostly type-specific SARS-CoV-2 response. We assessed antibody reactivity to nucleocapsid and spike antigens and correlated this IgG response to SARS-CoV-2 neutralization. We investigated the kinetics, breadth, magnitude, and level of cross-reactivity of IgG antibodies against SARS-CoV-2 and heterologous seasonal and epidemic coronaviruses at the clonal level in patients with mild or severe COVID-19 as well as in disease control patients. Little is known about the interplay between preexisting immunity to endemic seasonal coronaviruses and the development of a SARS-CoV-2–specific IgG response. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the cause of coronavirus disease 2019 (COVID-19). 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
